SATO Takeo
Associate Professor
Molecular basis of plant stress response via ubiquitin system
Department of Biological Sciences, Cell Structure and Function
| Theme | Molecular mechanism of plant adaptation to environmrntal stress via ubiquitin system |
| Field | Plant Physiology, Cell Biology, Biochemistry |
| Keyword | Signal transduction, Ubiquitin modification, Proteomics, Environmental stress, High CO2, Flowering, Plant-microbe interaction |
Introduction of Research
Ubiquitination is one of the fundamental post-translational modifications with ubiquitin, a conserved 76-amino acid protein present in eukaryotes. Ubiquitin forms several type of poly-ubiquitin chains to regulate protein degradation and subcellular localization, which plays critical roles in plant growth regulation in response to multiple environmental stresses. We previously identified a membrane localized ubiquitin ligase ATL31 regulating plant adaptation to carbon/nitrogen nutrient stress and defense response to pathogen attack. We are investigating biochemical and physiological significances of ubiquitin signals in plant stress adaptation.
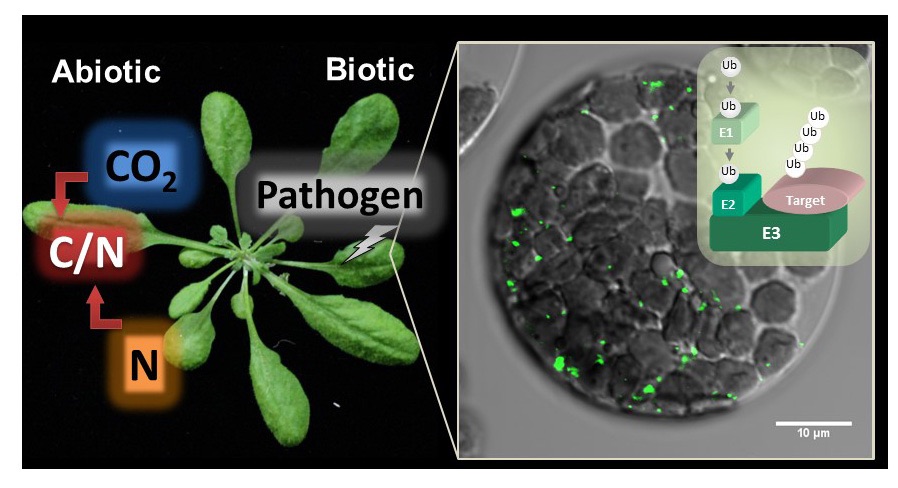 Plant adaptation to environmental stressess (C/N nutrient availability and pathogen attack)
Plant adaptation to environmental stressess (C/N nutrient availability and pathogen attack) Ubiquitin system
Ubiquitin system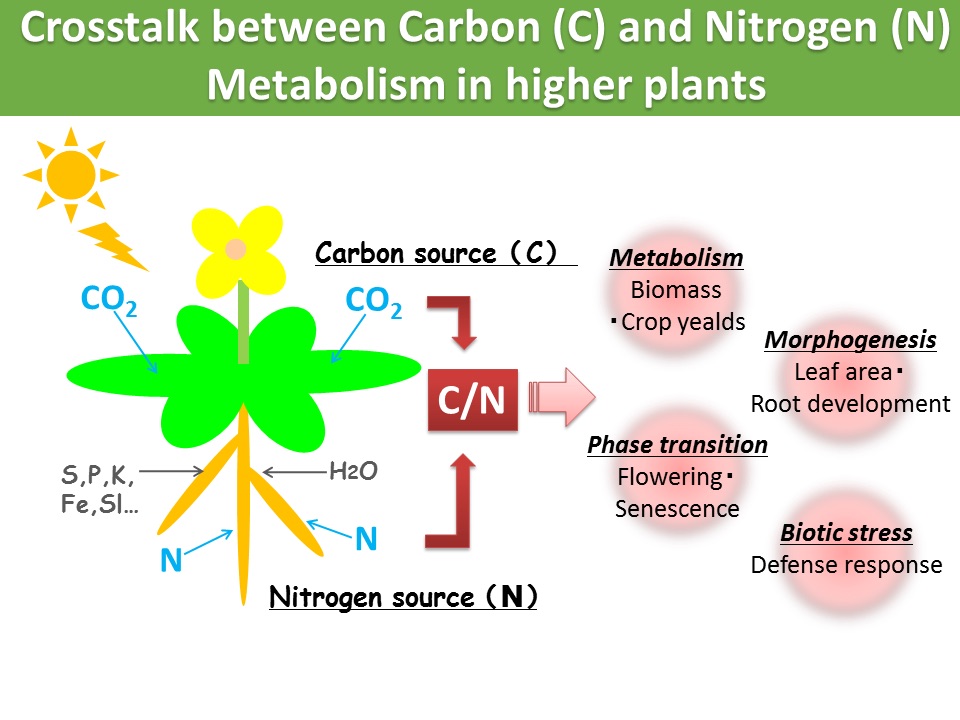 Plant growth regulation by C/N nutrient signal
Plant growth regulation by C/N nutrient signal CO2/N affects plant biomass and senescence progression
CO2/N affects plant biomass and senescence progression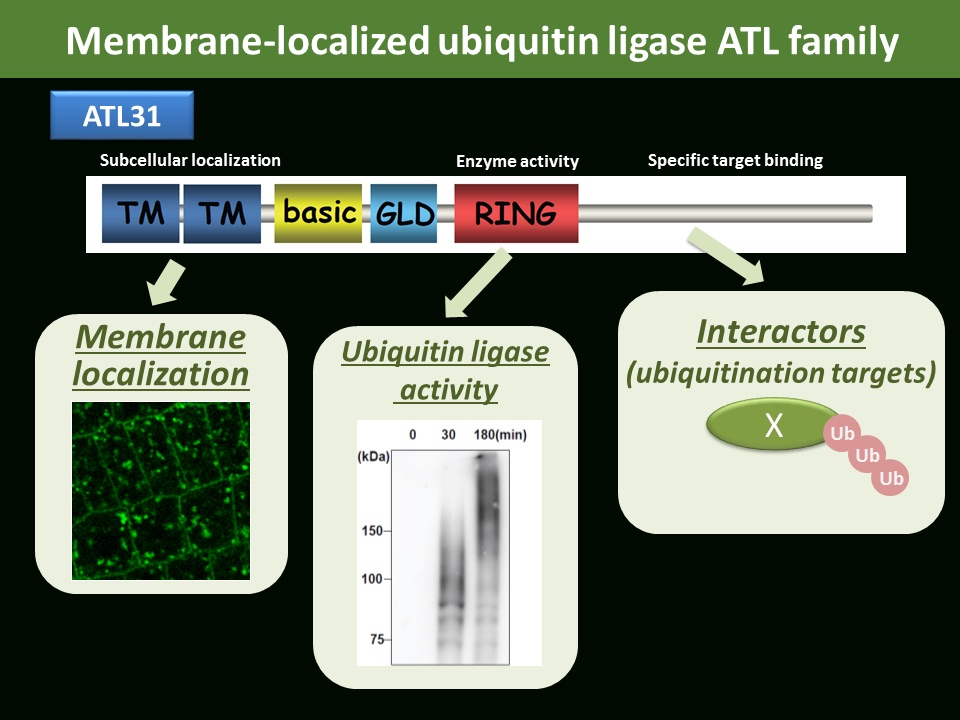 Membrane localized ubiqutiin ligase ATL31 in Arabidopsis
Membrane localized ubiqutiin ligase ATL31 in Arabidopsis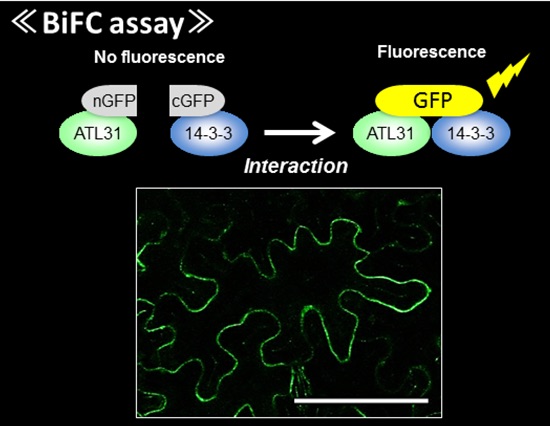 BiFC analysis to examine in vivo protein-protein interaction
BiFC analysis to examine in vivo protein-protein interactionRepresentative Achievements
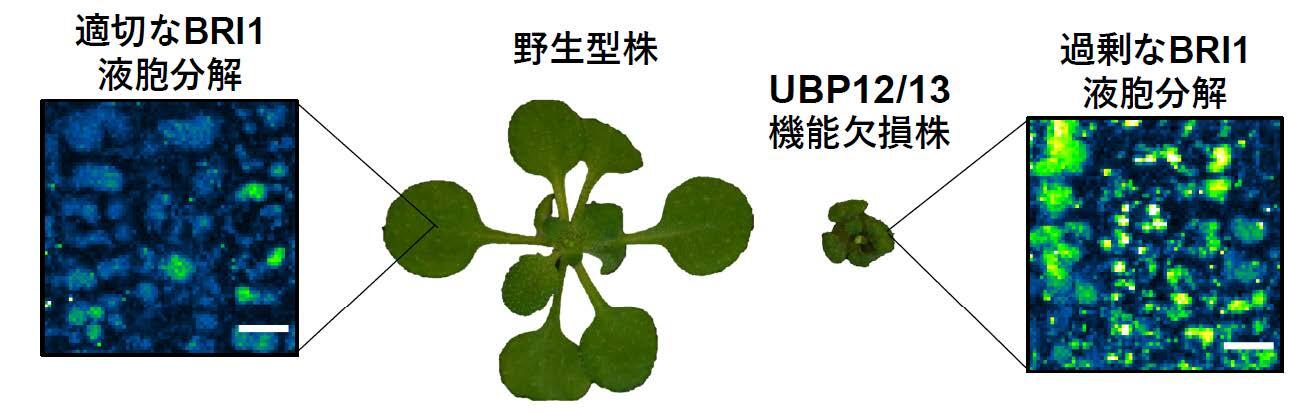
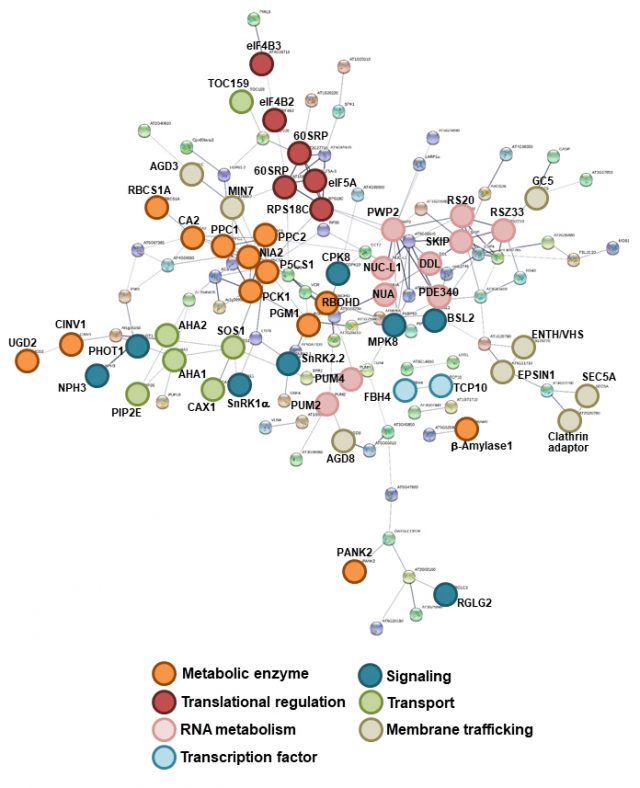
Deubiquitinating enzymes UBP12 and UBP13 limit stabilize the brassinosteroid receptor BRI1 (2022) Luo Y, Takagi J, Claus LAN, Zhang C, Yasuda S, Hasegawa Y, Yamaguchi J, Shan L, Russinova E and Sato T*, EMBO reports, 23: e53354.
The TGN/EE SNARE protein SYP61 and the ubiquitin ligase ATL31 cooperatively regulate plant responses to carbon/nitrogen conditions in Arabidopsis (2022) Hasegawa Y, Huarancca Reyes T, Uemura T, Baral A, Fujimaki A, Luo Y, Morita Y, Saeki Y, Maekawa S, Yasudaa S, Mukuta K, Fukao Y, Tanaka K, Nakano A, Takagi J, Bhalerao RP, Yamaguchi J and Sato T*, The Plant Cell, 34: 1354-1374.
Low nitrogen conditions accelerate flowering by modulating the phosphorylation state of FLOWERING BHLH 4 in Arabidopsis (2021) Sanagi M, Aoyama S, Kubo A, Lu Y, Sato Y, Ito S, Abe M, Mitsuda N, Ohme-Takagi M, Kiba T, Nakagami H, Rolland F, Yamaguchi J, Imaizumi T* and Sato T*, Proc. Natl. Acad. Sci. U. S. A., 118: e2022942118.
Protein phosphorylation dynamics under carbon/nitrogen-nutrient stress and identification of a cell death-related receptor-like kinase in Arabidopsis (2020) Li X, Sanagi M, -5-, Schulze WX, Regina F, Stitt M, Lunn JE, Nakagami H*, Sato T* and Yamaguchi J, Front. Plant Sci. 11: 377.
Arabidopsis CBL-Interacting Protein Kinases Regulate Carbon/Nitrogen-Nutrient Response by Phosphorylating Ubiquitin Ligase ATL31 (2017) Yasuda S, Aoyama S, Hasegawa Y, Sato T* and Yamaguchi J, Mol. Plant 10: 605-618.