ISHIMORI Koichiro
Professor
Molecular and Chemical Approaches to Elecidate Various Biological Processes Mediated by Biometals and Metalloproteins
Department of Chemistry, Physical Chemistry

| Theme | Structural and Functional Characterozations and Molecular Design of Biometals and Metalloproteins Based on Their Molecular Structures |
| Field | Bioinorganic Chemistry, Molecular Spectroscopy, Structural Biology, Physical Chemistry, Biophysics |
| Keyword | Metals in Biology, Heme/Porphyrin, Metalloproteins, Iron Metabolism in Cells, Mitochondrial Respiratory Chain, Electron Transfer, Nano Disc, Protein Folding |
Introduction of Research
All organisms have the specific transition metals, which are the essential elements to maintain their lives, although these metal contents are quite low in the body. Such indispensable functions of the transition metals are well described by the words of Prof. Negishi, one of the Nobel Laureates in Chemistry 2010, who have developed new fields of the organic chemistry utilizing the transition metals,
“Magical Power of d-Block Transition Metals”.
It should be noted here that primitive life had already known the "Magical Power" of metals and they have utilized it for the evolution. It is no exaggeration that life is maintained by the "Magical Power" of these transition metals, “biometals”, and the functional and structural characterization of “biometals” and proteins containing “biometals” will, therefore, pave the way for the comprehensive understanding of biological processes on the molecular level and contribute to reveal the molecular basis of life.
In several essential biometals, we have focused on iron, which is the most abundant transition metals and is utilized as the active centers for various kinds of matalloproteins essential for many biological processes. The major research projects in our laboratory are as follows:
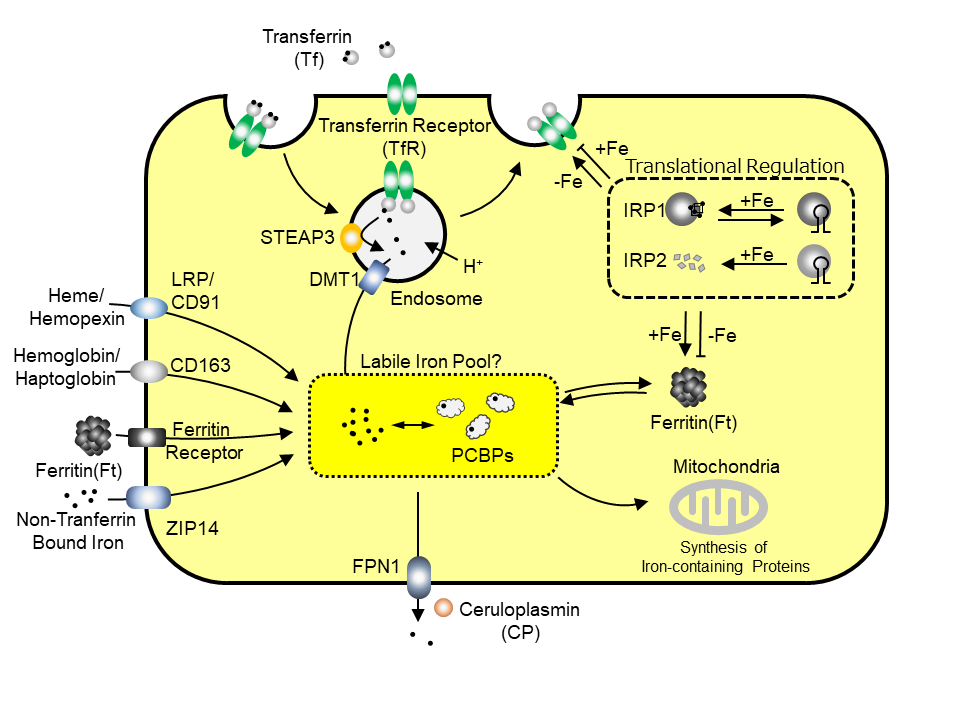
1. Molecular Regulation System for Iron Metabolism in Cells (Fig. 1)
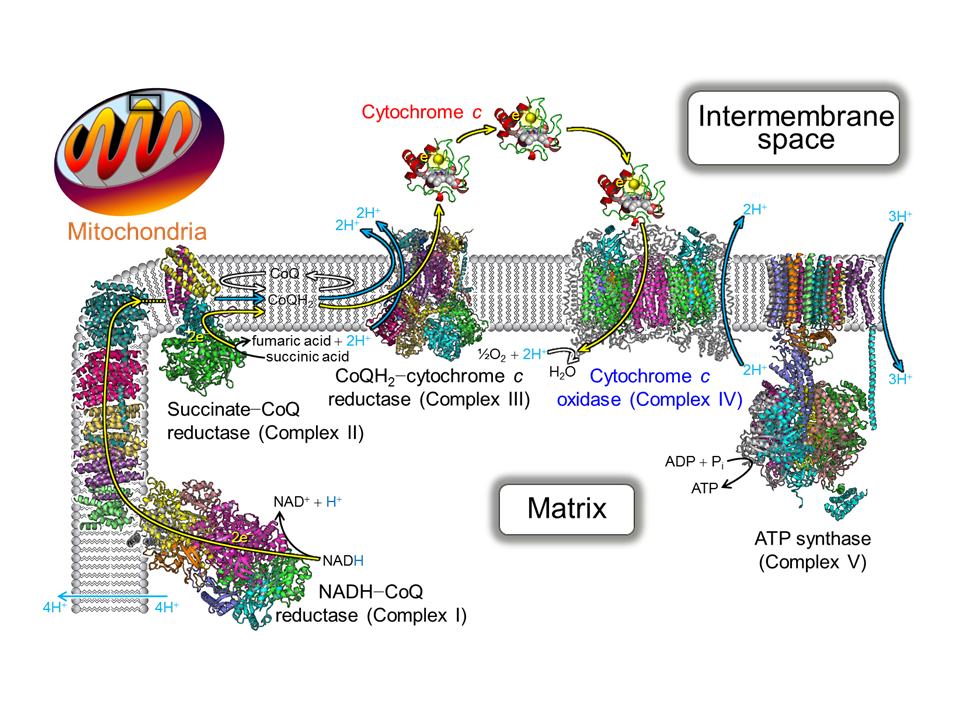
2. Regulation Mechanism of Electron Transfer in Respiratory Chain (Fig. 2)
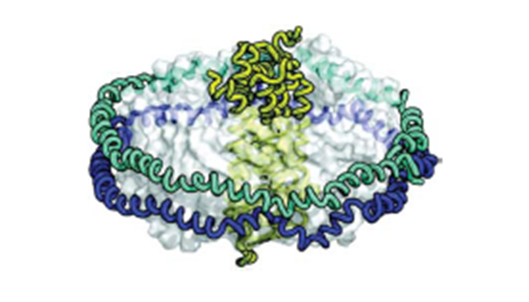
3. Structural and Functional Characterization of Nonodisc-inserted Membrane-bound Proteins (Fig. 3)
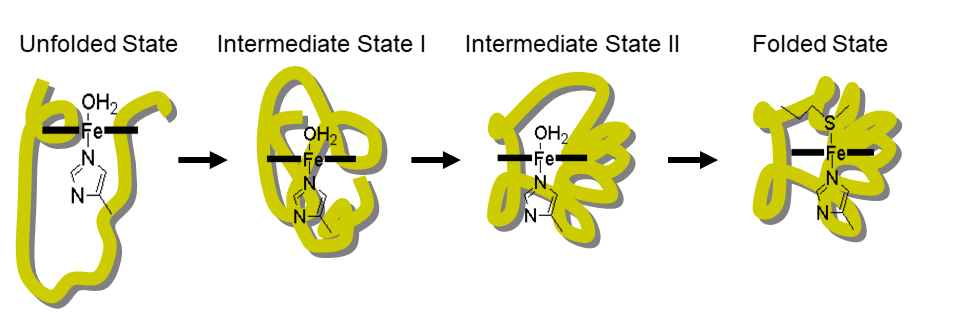
4. Molecular Mechanism for Protein Folding Utilizing Novel Physico-chemical Approaches (Fig. 4)
Based on these studies in “biometals” and biometal-containing proteins, biometals have been found to dynamically and systematically function as key elements in many crucial biological systems, and it has become evident that failure of the systems results in severe dysfunction of organs, which allows us to expect creation of new therapies and drug by controlling the "Magical Power".
Representative Achievements

Related industries
| Academic degree | Ph. D |
| Self Introduction | I was born in Kyoto, but not in the old central area, which would not fulfill the required conditions of the “traditional and genuine Kyoto people”. I started my academic carrier by studying one of the typical hemoproteins, hemoglobin, since more than thirty years ago. Before moving to Sapporo, I enjoyed a walk around Yoshida Hill and Shin-nyo-do Temple near the university. Now, walking around Maruyama Hill is my favorite, but it is getting harder to find time to go. |
| Academic background | 1984 B. S., Faculty of Engineering, Kyoto University 1986 M. S., Graduate School of Engineering, Kyoto University 1989 Ph.D., Graduate School of Engineering, Kyoto University 1989 Research Associate, Faculty of Engineering, Kyoto University 1995 Associate Professor, Graduate School of Engineering, Kyoto University 2005- Professor, Faculty of Science, Hokkaido University |
| Affiliated academic society | The Biophysical Society of Japan, The Japanese Biochemical Society, The Chemical Society of Japan |
| Project | Hokkaido University Ambitious Leader's Program (ALP) Ph.Discover |
| Room address | Science Building #7 7-105 |