Amyloid fibrils in Alzheimer's disease differently modulate sleep and cortical oscillations in mice depending on the type of amyloid
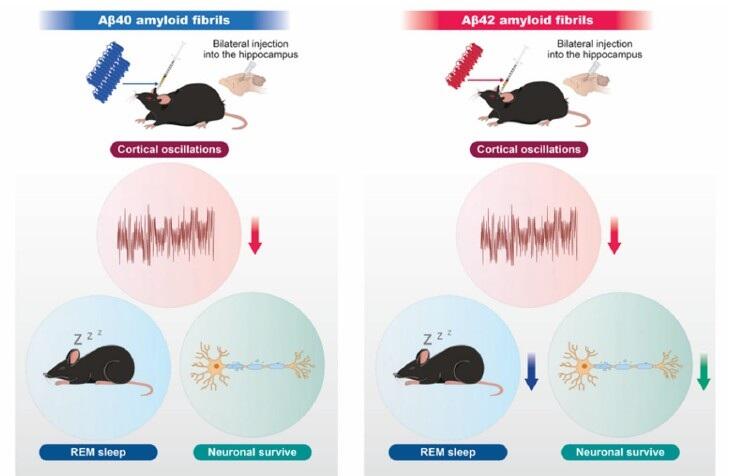
線維化Aβ40または線維化Aβ42をマウス海馬へ両側投与し、睡眠・覚醒状態及び皮質脳波活動(皮質振動)を解析した。両群で皮質脳波の変化が認められたが、線維化Aβ42ではレム睡眠の減少に加え、神経細胞脱落が示唆された。
Joint press release (in Japanese) by Hokkaido University and Tohoku University
Abstract
Alzheimer’s disease (AD) is characterized by aggregation and deposition of the amyloid-beta (Aβ) protein in patients’ brains, with aging contributing through oxidative stress and neuroinflammation. Sleep disturbances are common in patients with AD and exacerbate cognitive impairment. However, it remains unclear how aggregation of specific Aβ species in distinct brain regions contributes to sleep dysfunction. To address this, we characterized amyloid fibrils formed by Aβ1–40 (Aβ40) and Aβ1–42 (Aβ42) by assessing their structural and surface properties using fluorescence, CD, and NMR spectroscopy. Their morphology was also visualized using TEM and AFM. These analyses revealed that Aβ42 aggregates faster than Aβ40 and forms amyloid fibrils with distinct structural, surface, and morphological properties. To investigate their effects in vivo, we bilaterally injected Aβ40 and Aβ42 fibrils into the hippocampus of wild-type mice and recorded the electroencephalogram and electromyogram under freely moving conditions. Aβ42 amyloid fibrils significantly disrupted….
Read more on Biophysical Chemistry
Article Information:
Tomomi Sanagi, Masaki Okumura, Yuxi Lin, Shingo Kanemura, Eunyoung Moon, Yunseok Heo, Keiko Takahara, Young-Ho Lee, Tomomi Tsunematsu, Amyloid fibrils in Alzheimer’s disease differently modulate sleep and cortical oscillations in mice depending on the type of amyloid, Biophysical Chemistry, 333, 2026
DOI:10.1016/j.bpc.2026.107599