MIURA Atsushi
Associate Professor
Chemical Photonics: Development and application of novel analytical method using photon pressure as local perturbation
Department of Chemistry, Inorganic and Analytical Chemistry

| Theme | Construction of a mesoscopic reaction/measurement space/field by using a photon pressure as a local perturbation. Exploration and elucidation of novel physical/chemical phenomena induced in the mesoscopic by the photon pressure and its mechanism. Development and application of high-sensitivity next-generation analytical methods based on above mentioned themes. |
| Field | Analytical chemistry, Photochemistry, Chemical Photonics |
| Keyword | Laser trapping/Optical tweezers, Microscopy/Spectroscopy, Crystallization, Phase transition, Phase separation, Self-assembly |
Introduction of Research
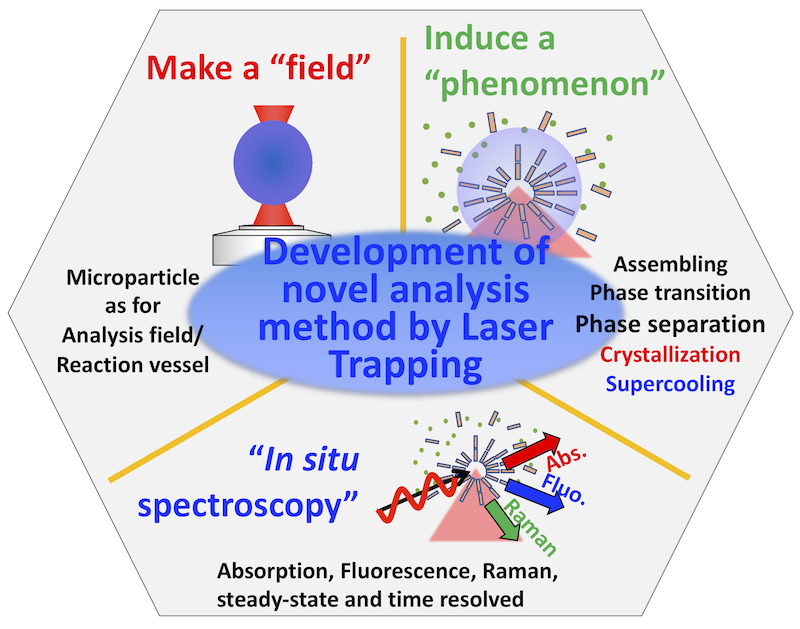
A simple, in situ, and highly sensitive and accurate analytical/measurement method for trace substances is useful and demanded in all fields. By utilizing light, especially a photon pressure, as a local perturbation, we can construct a mesoscopic reaction/measurement space/field. We explore novel chemical/physical phenomena induced by the photon pressure and utilize it to carry out simultaneous and high sensitivity optical measurement by light. We are working on the development of next-generation analytical methods for "measurement" (Figure 1). We are creating a mesoscopic space/field by laser trapping/optical tweezers technique, which can be used for the analysis, inducing reaction/accumulation/self-assembly and so on. We study such phenomena by using various optical microscope and microspectroscopy techniques Our recent activities are as follows;
1) Measurement and clarification of "intrinsic" liquid properties of a single aerosol droplet
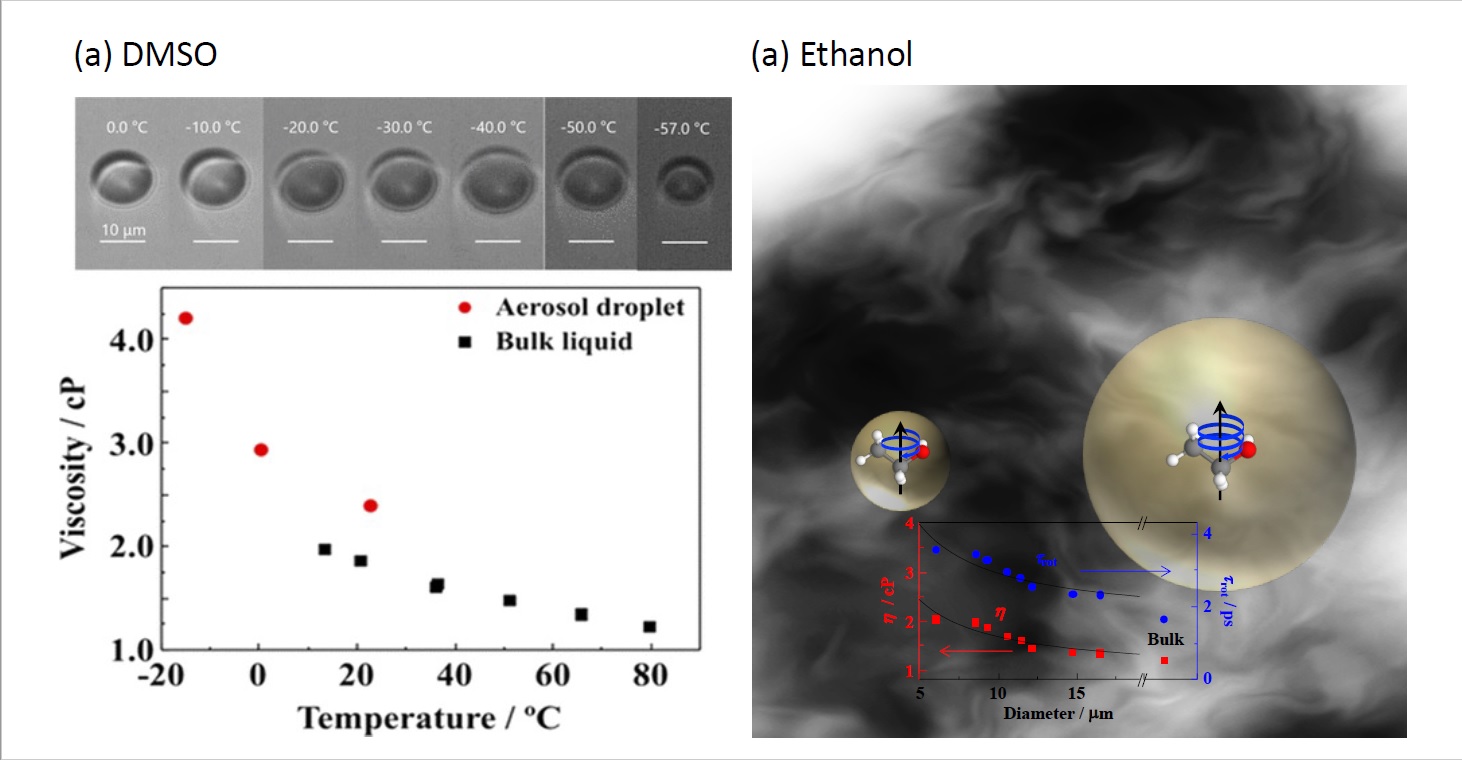
It is known as common knowledge that the freezing point of water is 0°C. However, microscopic water droplets (aerosols) on the micrometer scale levitated in the air do not freeze until extremely low temperatures (below -50°C) and exist as liquids (supercooled liquids). In other words, the physical properties of liquids are different from what we know as "common knowledge ". Recently, we have clarified for the first time in the world that even organic liquids other than water easily become supercooled liquids by aerosolization and do not freeze until much lower temperatures than the freezing point(Fig. 2a), and that the viscosity of aerosol droplets changes depending on the droplet size (Fig. 2b). Currently, we are working on elucidating the mechanism of aerosol droplet properties that differ from "common knowledge.
2) Developing an ultra-trace analysis method using photo-induced single polymer particles as an analytical field.
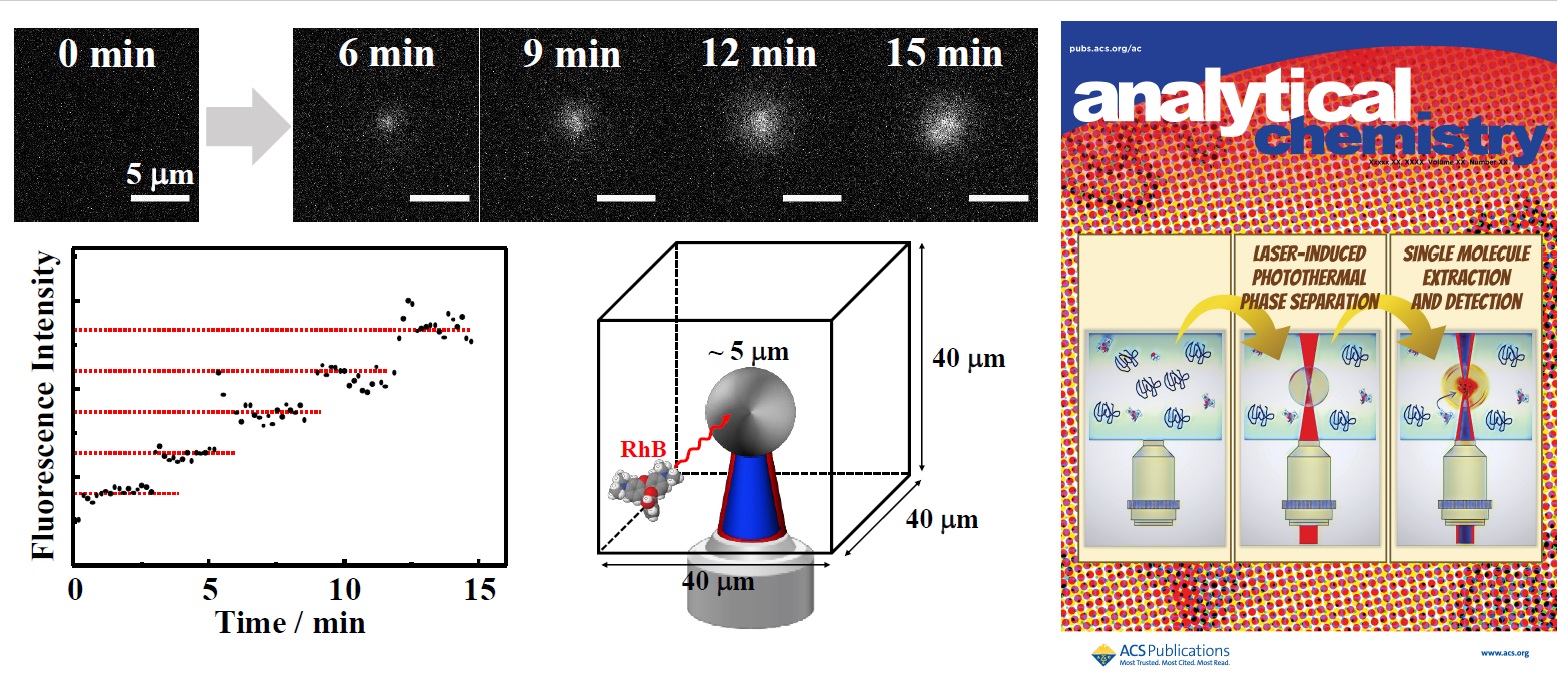
When a temperature-responsive polymer, poly-N-isopropylacrylamide (PNIPAM), is irradiated with a focused 1064-nm laser beam into an aqueous mixture of alcohols, phase separation is induced by localized photothermal conversion, resulting in the formation of a single particle (~10 µm). We have recently shown that extremely dilute solute molecules dispersed in solution (concentration: 10-15 M level) can be extracted and detected at the single-molecule level with ultra-high sensitivity by using this polymer particle as a field for separation and extraction analysis (Fig. 3). Extraction analysis at the single-molecule level is an unexplored field. We are conducting basic research on the application of these results as an analytical method for trace biomolecules.
3) Development of simple and easy protein crystallization method using laser trapping
Since the properties of proteins are closely correlated with their structure, it is important to clarify 3D structure of protein molecule. However, it is very difficult to crystallize proteins, which are large biological macromolecules. I have been developing a simple and easy method for protein crystallization using laser trapping. From the recent results obtained with a lysozyme, we have succeeded in rapid and efficient crystal formation and growth by inducing a liquid-liquid phase separation in a protein solution by laser trapping (Fig. 3). This laser trapping-induced crystallization method is expected to be useful for easy crystallization of various protein molecules.
Representative Achievements
A. Miura, R. Nohara, A. Nojima, N. Kitamura, Anal. Chem., 93, 3202-3208 (2021).
A. Miura, R. Nakajima, S. Abe, N. Kitamura, J. Phys. Chem. A, ASAP (2020).
A. Miura, Y.-H. Huang, H. Masuhara, Appl. Phys. B-Lasers Opt., 112, 473-477 (2013).
K. Sano, A. Miura, S. Yoshii, M. Okuda, M. Fukuta, Y. Uraoka, T. Fuyuki, I. Yamashita, K. Shiba, Langmuir, 29, 12483-12489 (2013).
| Academic degree | Ph. D |
| Self Introduction | Science Building 7 |
| Academic background | 1995 B.S. Department of Chemistry, Faculty of Science, Hokkaido University (Japan) 1997 M. ES Graduate School of Environmental Science, Hokkaido University (Japan) 2001 Ph.D Graduate School of Science, Kwansei Gakuin University (Japan) 2001 Postdoctoral research fellow, Department of Chemistry, Katholiek University Leuven (Belgium) 2004 Postdoctoral research fellow, Nara Institute for Science and Technology (Japan) 2008 Associate Researcher, Department of Applied Chemistry, Faculty of Science, National Chiao Tung University (Taiwan) 2008 Visiting researcher, Nara Institute for Science and Technology (Japan) 2009 Assistant Professor, Department of Applied Chemistry, Faculty of Science, National Chiao Tung University (Taiwan) 2014 – Associate Professor, Department of Chemistry, Faculty of Science, Hokkaido University (Japan) |
| Affiliated academic society | The Chemical Society of Japan, The Japan Society for Analytical Chemistry, The Japan Society of Applied Physics, The Japanese Photochemistry Association, The Society of Polymer Science |
| Room address | Science Building 7 03-08 |